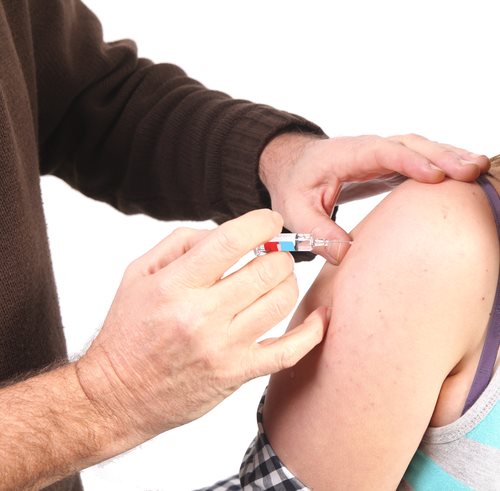
On October 15, 2012, the National Institutes of Health (NIH) announced funding that will help researchers find out if a higher dose of a vaccine for pneumonia can create a stronger immunity for older adults who received similar vaccines in the past.
According to the NIH, the new study will compare results from vaccines given to children between 6 weeks and 5 years old and adults 50 years of age or older. About 882 men and women between 55 and 74 have elected for the study.
The study addresses one of the largest health epidemics in the United States. According to the Centers for Disease Control and Prevention, over 300,000 people in the U.S. are hospitalized every year because of pneumonia. Certain types of pneumonia are particularly dangerous.
S. pneumoniae can lead to the upper respiratory tract becoming infected, and the infection can spread to the lungs, blood, middle ear, and even the nervous system. Children under the age of 5 and adults over 65 are the most likely to contract pneumococcal pneumonia, and it is easier to become re-infected once you were originally infected.
Because of the severity of the illness, pneumonia currently ranks as the 15th leading cause of death in the U.S. Adults over the age of 55 account for 92 percent of pneumonia deaths every year.
The study is occurring at six National Institute of Allergy and Infectious Diseases (NIAID) vaccine and treatment units in across the county. The following centers are conducting the study: University of Iowa, Saint Louis University, Cincinnati Children’s Hospital Medical Center—Infectious Diseases, Vanderbilt University Vaccine Research Center, Baylor College of Medicine, and the Seattle Group Health Research Institute.
The hope is to successfully vaccinate adults who have received lower dosages of the vaccine in the last 3 to 7 years.
Source: National Institutes of Health